It also may result from the combining of radiation from a number of regions which are far from each other.īroadening due to local effects Natural broadening Broadening due to extended conditions may result from changes to the spectral distribution of the radiation as it traverses its path to the observer. Broadening due to local conditions is due to effects which hold in a small region around the emitting element, usually small enough to assure local thermodynamic equilibrium. These reasons may be divided into two general categories – broadening due to local conditions and broadening due to extended conditions. There are several reasons for this broadening and shift. In addition, its center may be shifted from its nominal central wavelength. A spectral line extends over a range of frequencies, not a single frequency (i.e., it has a nonzero linewidth). There are a number of effects which control spectral line shape. These series were later associated with suborbitals. These series exist across atoms of all elements, and the patterns for all atoms are well-predicted by the Rydberg-Ritz formula. Originally all spectral lines were classified into series: the principal series, sharp series, and diffuse series. Many spectral lines of atomic hydrogen also have designations within their respective series, such as the Lyman series or Balmer series. More detailed designations usually include the line wavelength and may include a multiplet number (for atomic lines) or band designation (for molecular lines). 
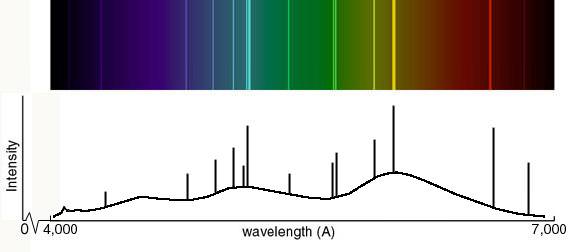
Neutral atoms are denoted with the Roman numeral I, singly ionized atoms with II, and so on, so that, for example, Fe IX represents eight times ionized iron. In other cases, the lines are designated according to the level of ionization by adding a Roman numeral to the designation of the chemical element. Strong spectral lines in the visible part of the spectrum often have a unique Fraunhofer line designation, such as K for a line at 393.366 nm emerging from singly-ionized Ca +, though some of the Fraunhofer "lines" are blends of multiple lines from several different species. Spectral lines also depend on the temperature and density of the material, so they are widely used to determine the physical conditions of stars and other celestial bodies that cannot be analyzed by other means.ĭepending on the material and its physical conditions, the energy of the involved photons can vary widely, with the spectral lines observed across the electromagnetic spectrum, from radio waves to gamma rays. Several elements, including helium, thallium, and caesium, were discovered by spectroscopic means. Spectral lines are highly atom-specific, and can be used to identify the chemical composition of any medium. The intensity of light, over a narrow frequency range, is increased due to emission by the hot material. By contrast, a bright emission line is produced when photons from a hot material are detected, perhaps in the presence of a broad spectrum from a cooler source. The intensity of light, over a narrow frequency range, is reduced due to absorption by the material and re-emission in random directions. An absorption line is produced when photons from a hot, broad spectrum source pass through a cooler material. 
Which type of line is observed depends on the type of material and its temperature relative to another emission source. Then the energy will be spontaneously re-emitted, either as one photon at the same frequency as the original one or in a cascade, where the sum of the energies of the photons emitted will be equal to the energy of the one absorbed (assuming the system returns to its original state).Ī spectral line may be observed either as an emission line or an absorption line. When a photon has about the right amount of energy (which is connected to its frequency) to allow a change in the energy state of the system (in the case of an atom this is usually an electron changing orbitals), the photon is absorbed. Spectral lines are the result of interaction between a quantum system (usually atoms, but sometimes molecules or atomic nuclei) and a single photon. 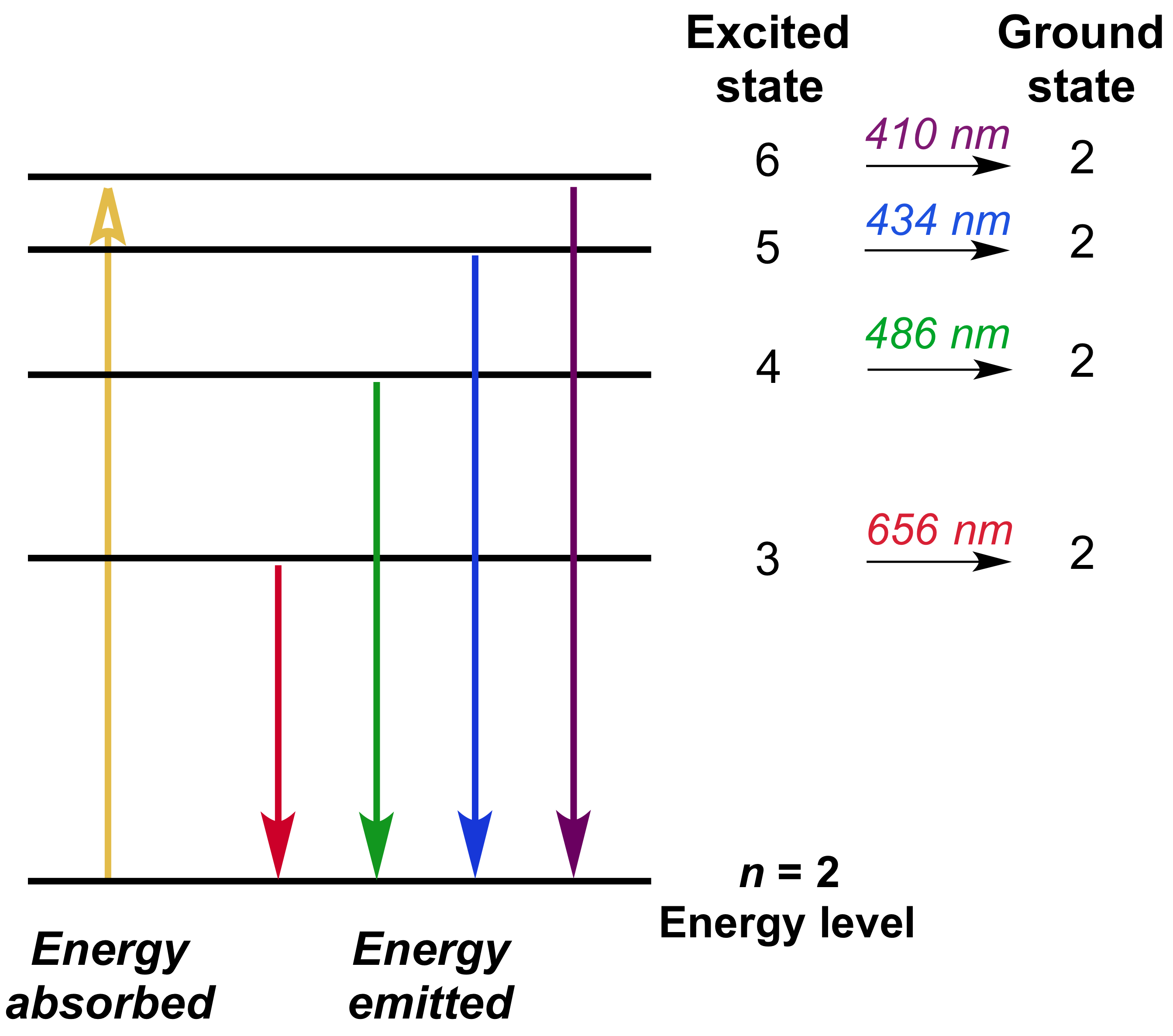
Continuous spectrum of an incandescent lamp (mid) and discrete spectrum lines of a fluorescent lamp (bottom)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
